Clozapine-associated pericarditis and pancreatitis in children and adolescents: A systematic literature review and pharmacovigilance study using the VigiBase database - ScienceDirect
Por um escritor misterioso
Descrição

Drug induced pancreatitis is the leading known cause of first attack acute pancreatitis in children - ScienceDirect

Protocol for Clozapine Rechallenge in a Case of Clozapine-Induced Myocarditis - G. Shivakumar, N. Thomas, M. Sollychin, A. Takács, S. Kolamunna, P. Melgar, F. Connally, C. Neil, C. Bousman, M. Jayaram, C.

PDF) Clozapine-associated pericarditis and pancreatitis in children and adolescents: A systematic literature review and pharmacovigilance study using the VigiBase database

Evidence‐based clinical practice guidelines for acute pancreatitis: proposals - Mayumi - 2002 - Journal of Hepato-Biliary-Pancreatic Surgery - Wiley Online Library
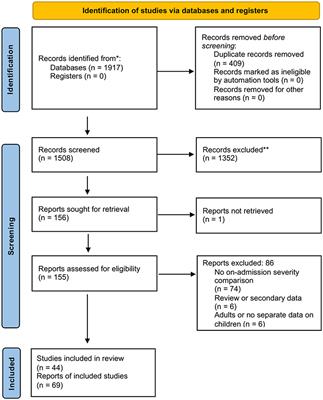
Frontiers Admission risk factors and predictors of moderate or severe pediatric acute pancreatitis: A systematic review and meta-analysis

1410344 PDFs Review articles in ADOLESCENTS

PDF) Clozapine-associated pericarditis and pancreatitis in children and adolescents: A systematic literature review and pharmacovigilance study using the VigiBase database

PDF) Impact of education and continuous professional development in Portuguese Pharmacy Technicians attitude's to adverse drug reaction reporting system

Asparaginase-associated pancreatitis in childhood acute lymphoblastic leukaemia: an observational Ponte di Legno Toxicity Working Group study - The Lancet Oncology
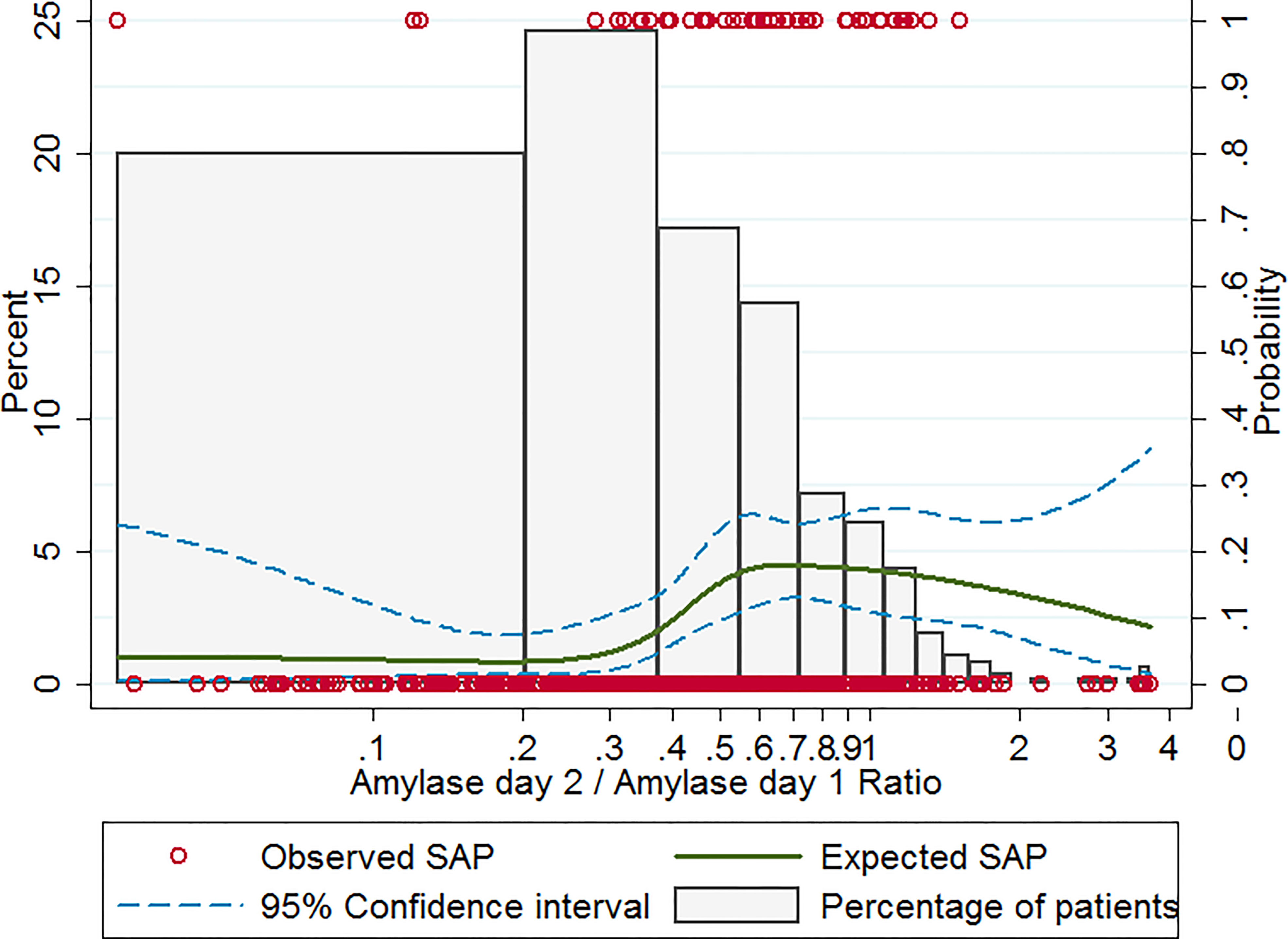
Frontiers Non-linear correlation between amylase day 2 to day 1 ratio and incidence of severe acute pancreatitis

Antipsychotic drugs - ScienceDirect

Colchicine for children with pericarditis: systematic review of clinical studies

PDF) Clozapine-associated pericarditis and pancreatitis in children and adolescents: A systematic literature review and pharmacovigilance study using the VigiBase database

PDF) Impact of education and continuous professional development in Portuguese Pharmacy Technicians attitude's to adverse drug reaction reporting system